
 |
|||||||||||||||
CO2 concentrating mechanisms (CCMs) interally elevate CO2 at the active site of Rubisco (see my Rubisco research page). The best known systems are C4 and CAM photosynthesis, where CO2 in temporarily captured and attached to small sugar and later released around Rubisco where it is assimilated via the Calvin Cycle (of the standard C3 photosynthetic pathway). This process uses more energy than C3 photosynthesis but it reduces or eliminatates photorespiration and can improve water ant nitrogen use efficiency (again see my Rubisco page). However, there two other functional classes of CCMs that are not known as well. Carboxysome and pyrenoid-based CCMs do not make temporary sugars, but instead they directly pump CO2 and bicarbonate to a special region within the cell where all the Rubisco is located. This is also an energy consuming process that has the same effect of increasing CO2 aroung Rubisco.
Cyanobacteria (and some photosynthetic proteobacteria) have small structures within their cells called carboxysomes. These often appear hexagonal in cross-section and encapsulate all the cell's Rubisco wihtin a protein shell. Several groups have demonstrated that the carboxysome is a critical structure for proper CCM function. The discovery of genes involved in CCM function has been facilitated by the inducible/supressable nature of many carboxysome-based systems. At very high CO2 (generally tested at 1% CO2) the CCM is down-regulated and can be turned back on within hours of returning to ambient CO2. Another recent suprise was the realization that there are two types of carboxysomes with unrelated genes (sequences are not similar, but functions are). The normal disjunct is between cyanobacterial and proteobacterial carboxysomes, however the open ocean cyanobacterium Synechococcus WH8102 has the protoebaccterial form (see Badger, Hanson, and Price 2002). This cyanobacterium is involved in large open ocean blooms that have a major impact on oceanic carbon sequestration. We are currently studying the physiology of this unusual organism to determine how a proteobacterial carboxysome functions in a cyanobacterial cell.
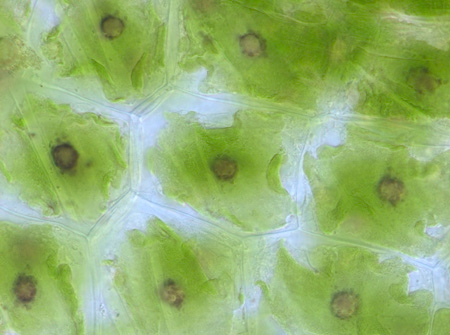
 Some
hornworts and algae (see my bryophyte reserach page)
have a structure, called the pyrenoid, that is functionally analagous to the
carboxysome in cyanobacteria. This structure is located in the chloroplast and
contains all the Rubisco (like a carboxysome), however, it is not enclosed in
a protein shell and it is often traversed by thylakoid memberanes. The presence
of this structure has been correlated with CCM funtion in hornworts (see Hanson
et al. 2002) and demonstrated more definitively in some algae like Chlamydomonas
reinhardtii, where many genes involved in CCM function have been identified.
My lab studies variation in pyrenoid structure and function primarily within
hornworts (the only land plants with pyrenoids) but also in Chlamydomonas
through collaborations with other labs around the world.
Some
hornworts and algae (see my bryophyte reserach page)
have a structure, called the pyrenoid, that is functionally analagous to the
carboxysome in cyanobacteria. This structure is located in the chloroplast and
contains all the Rubisco (like a carboxysome), however, it is not enclosed in
a protein shell and it is often traversed by thylakoid memberanes. The presence
of this structure has been correlated with CCM funtion in hornworts (see Hanson
et al. 2002) and demonstrated more definitively in some algae like Chlamydomonas
reinhardtii, where many genes involved in CCM function have been identified.
My lab studies variation in pyrenoid structure and function primarily within
hornworts (the only land plants with pyrenoids) but also in Chlamydomonas
through collaborations with other labs around the world.
Proper CCM function requires careful spatial and temporal arrangement of cells or enzymes. This is most evident when considering CO2 diffusion since elevation of CO2 must occur at the active site of Rubisco and these CO2 molecules must be prevented from leaking away from Rubisco prior to catalysis (if not, a futile cycle will be created). The structural arrangement is hardest to understand at the subcellular level, especially with a pyrenoid-based system since there is no bounding membrane or protein coat. There have been theories about the arrangement of the enzyme carbonic anhydrase (which interconverts CO2 and bicarbonate) and Rubisco or PSII (see Hanson et al. 2003) along with other hypotheses of structure/function relationships in pyrenoids (see Hanson et al 2002), but much more data are needed. In my lab, we are primarily comparing hornworts with variable pyrenoid forms to answer how structural variation affects function. I am also interested in learning the extent to which linear electron transport from photosystem II (PSII) or cyclic electron transport around photosystem I (PSI) are involved in providing the energy needed for CCM function under various enviromental conditions. We are currently developing a membrane inlet mass spectrometery system here at UNM (see my isotopes in photosythesis page) and fluorescence methods (see my fluorescence studies of photosynthesis page) to test our hypotheses.
As mentioned above, some organisms can induce and suppress their CCMs in response to varying environmental conditions. We have been collaborating with Dr. Nate McDowell of the Earth and Environmental Sciences division of Los Alamos National Laboratories to develop methods for monitoring physiological aspects of the induction process in real-time. This is based on a new piece of equipment called a Tunable Diode Laser (TDL) which can measure the isotopic composition of CO2 in an airstream in real-time (see my isotopes in photosythesis page). There are two stable isotopomers of CO2 in our atmosphere and Rubisco prefers to use the lighter one. When a CCM is functioning Rubisco is forced to use more of the heavy isotopomer than without a CCM, so monitoring the relative usage of the lighter isotope can tell us about the activity of the CCM.